
Source: PaxForex Premium Analytics Portal, Fundamental Insight
The countdown has started, and we are not talking about presidential elections in the USA. The whole world is waiting for the results of the coronavirus vaccine testing, which is in the late testing stage.
Positive clinical outcomes should direct to relatively rapid FDA approval for emergency use (EUA). It is possible that within the next few months, safe and effective vaccines will be available throughout the United States. It could be a turning point in the fight against a pandemic that has so far taken more than 200,000 lives across the country and seriously undermined the economy.
Pfizer is a U.S. company that is likely to first announce the results of testing of its respective vaccine candidates at a later stage. But what if the results are published late? Believe it or not, delays can be great news.
On July 27, 2020, Pfizer and its partner, the German biotechnology company BioNTech, began Phase 3 of the clinical trial of a candidate for the BNT162b2 vaccine. The businesses stated back then that they could apply for regulatory inspection in October if the tests were successful.
On September 12, Pfizer and BioNTech suggested extending the last phase to include 44,000 participants rather than the initially scheduled 30,000 patients. Nonetheless, this did not affect their expected schedule. Pfizer and BioNTech confirmed that they "still expect the final performance data to be available by the end of October.
There are two other drug manufacturers with candidates for the vaccine that is in the late stages of testing in the U.S. - AstraZeneca and Johnson & Johnson. However, testing of AstraZeneca in the U.S. has still been suspended due to a severe reaction with the company's clinical trial participant in the UK. J&J started Phase 3 testing of its candidate for the COVID-19 vaccine less than two weeks ago and anticipates results only in early 2021.
You may think that any delays for Pfizer will automatically be bad news - but there is a chance that potential delays could be great news.
How? It all comes down to what the FDA will require to provide EUA.
The FDA is going to release a directory on the criteria that the vaccine must meet shortly to obtain emergency use approval. Although the details are not yet known, there have been leaks about some potential provisions.
Some of those criteria are specifically noteworthy. The Food and Drug Administration (FDA) is likely to require participants in late clinical trials of candidates for the vaccine to be monitored for two months after receiving a second dose of the vaccine. Besides, the agency is expected to require placebo groups participating in the studies to have at least five cases of severe COVID-19.
Pfizer can track how it fulfills the first of these two criteria, but if the placebo group does not have enough participants who develop severe cases, they will not be able to apply to the EUA.
If such a scenario happens, it will mean that almost all non-vaccinated individuals who participated in late studies will not be infected with the coronavirus. It would be unbelievable and would show substantial progress in the impact of COVID-19 on the country. It would also indicate that Pfizer would have more vaccinated participants to evaluate the safety profiles of their vaccines.
It would improve Americans' trust in the vaccines.
Of course, there is also the chance that Pfizer may defer reporting results at a later stage for internal grounds, like the necessity to get more time for data collection and analysis. This scenario does not necessarily lead to a decline in stock prices, although it is likely to affect the company one way or another.
Chances are almost certainly high that Pfizer will reach the announced deadlines for late submission of results for its vaccine candidates. It would be almost miraculous if very few participants in placebo groups would not have severe COVID-19 cases.
It should be remembered that the company also knows what is at stake. Crucial internal errors that may delay the announcement of their outcomes should be doubtful.
The companies will get $1.95 billion from the U.S. government to deliver 100 million doses of BNT162b2 in case the vaccine obtains EUA. By the way, the contract could be extended by an extra 500 million doses.
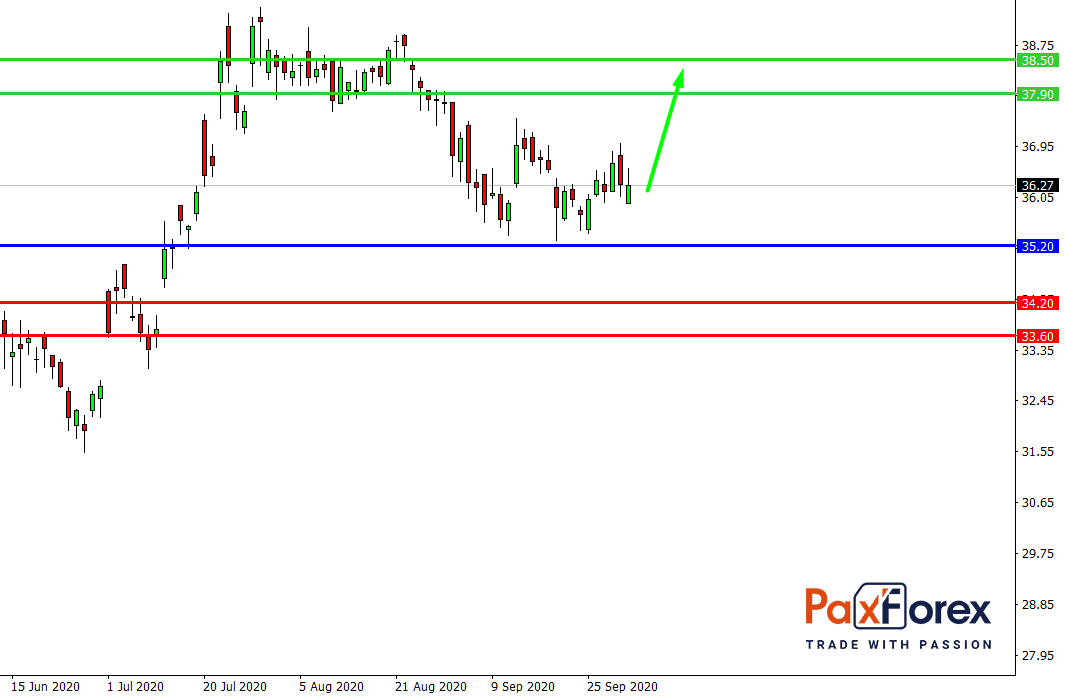
Provided that the company is traded above 35.20, follow the recommendations below:
- Time frame: D1
- Recommendation: long position
- Entry point: 36.38
- Take Profit 1: 37.90
- Take Profit 2: 38.50
Alternative scenario:
In case of breakdown of the level 35.20, follow the recommendations below:
- Time frame: D1
- Recommendation: short position
- Entry point: 35.20
- Take Profit 1: 34.20
- Take Profit 2: 33.60













